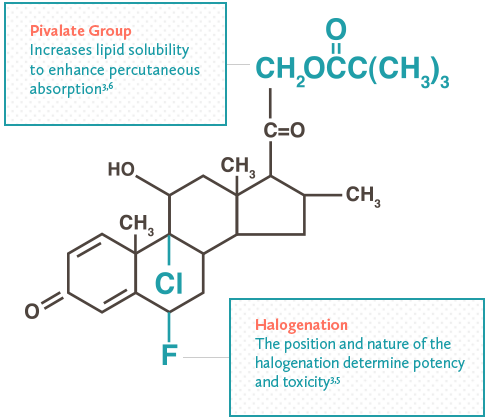
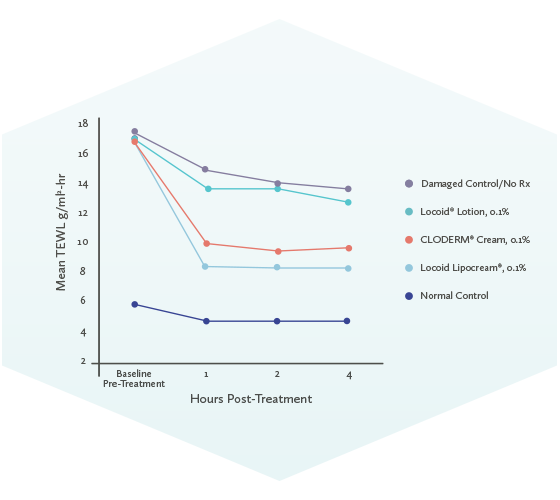
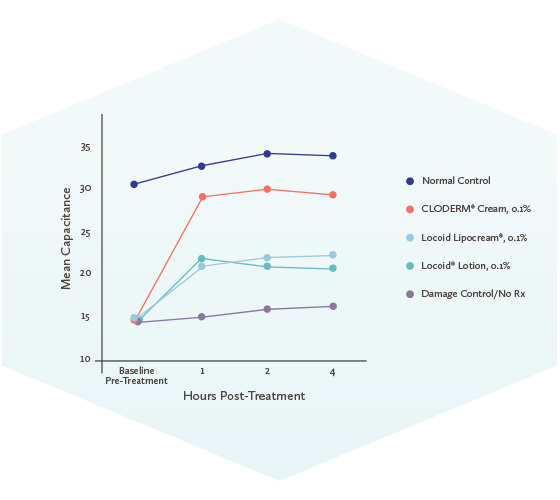
Patient Instructions: Redeem this coupon ONLY when accompanied by a valid prescription for CLODERM® Cream, 0.1% or Clocortolone Pivalate Cream, 0.1%. Commercially insured patients pay as little as $0 for their prescription. Maximum reimbursement limits apply.
This coupon is good for up to 12 uses and is not transferable. This coupon offer varies by size.
Pharmacist instructions for a patient with an Eligible Third Party Payer: Submit the claim to the primary Third Party Payer first, then submit the balance due to Change Healthcare as a Secondary Payer COB [coordination of benefits] with patient responsibility amount and a valid Other Coverage Code (eg, 8). The patient-pay amount will be reduced up to the maximum per size and reimbursement will be received from Change Healthcare.
For Patients requiring Prior Authorization: Submit the claim from the primary Third Party Payer to Change Healthcare as a secondary payer COB with patient responsibility amount and valid Other Coverage Code (eg, 03). The patient pay amount will be reduced by up to the maximum per size and reimbursement will be received from Change Healthcare.
Valid Other Coverage Code required:
For any questions regarding Change Healthcare online processing, please call the Help Desk at 1-800-422-5604.
For offer details and patients with questions, please call 1-866-259-4448.
Please consult full prescribing information for CLODERM® Cream. Not valid for patients reimbursed by federal health care programs, including Medicare, Medicaid, Tricare, the Department of Veterans Affairs, state maternal and child health block grant programs under 42 U.S.C. 701 et. seq. state social service block grant programs under 42 U.S.C. section 1397 et. seq. or any other similar federal or state health care program. Void where prohibited by law, taxed or restricted. Void outside the United States. Patient is responsible for reporting receipt of card program rewards to any private insurer that pays for or reimburses any part of the prescriptions filled with this card. Void if reproduced. It is illegal for any person to sell, purchase, or trade, or offer to sell, purchase or trade, or to counterfeit this card.
EPI Health reserves the right to rescind, revoke or amend this offer at any time without notice.
GMP library not installed, please contact server admin.